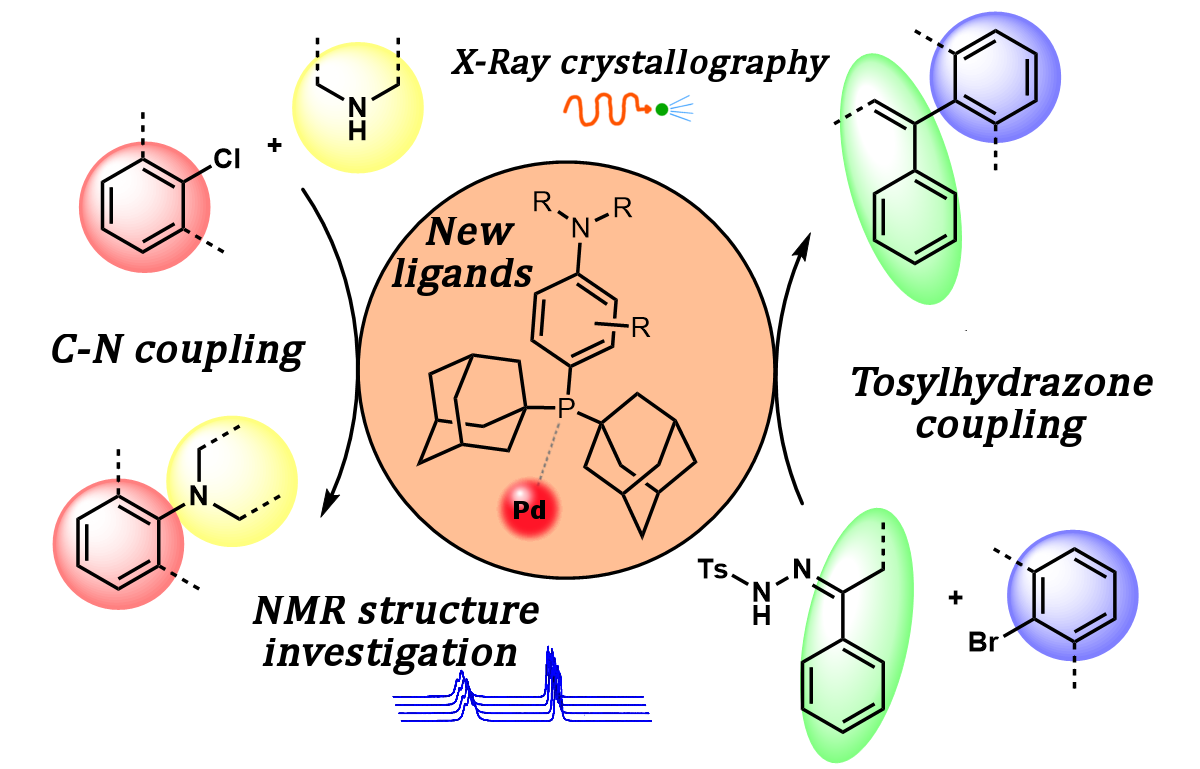
76. Synthesis, Structural Analysis and Application of Aryl‐Diadamantyl Phosphine ligands in Palladium Catalyzed Cross‐Coupling Reactions
Synthesis, Structural Analysis and Application of Aryl‐Diadamantyl Phosphine ligands in Palladium Catalyzed Cross‐Coupling Reactions, Ádám Sinai, Dániel Csaba Simkó, Fruzsina Szabó, Attila Paczal, Tamás Gáti, Attila Bényei, Zoltán Novák, András Kotschy, Eur. J. Org. Chem. 2020, 9, 1122-1128. DOI: 10.1002/ejoc.201901834 | [Full Text Link] [Supp. Info. Link]
Synthesis, temperature dependent NMR structure investigation and utilization of a new, stable and easily accessible aryl‐diadamantylphosphine ligand family is reported. The bulky and electron rich phosphorous center of the ligand enhances the catalytic activity of palladium in cross‐coupling reactions of sterically demanded ortho substituted aryl halides. In our study we demonstrated the synthetic applicability of the new phosphine ligands in Buchwald‐Hartwig and tosyl hydrazone coupling reactions.
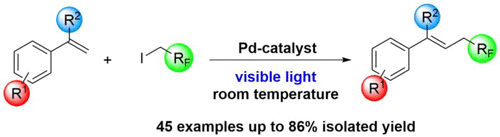
77. Photocatalytic Palladium-Catalyzed Fluoroalkylation of Styrene Derivatives
Photocatalytic Palladium-Catalyzed Fluoroalkylation of Styrene Derivatives, Réka Adamik, Tamás Földesi, Zoltán Novák, Org. Lett. 2020, 22, 8091-8095. DOI: 10.1021/acs.orglett.0c03043 | [Full Text Link] [Supp. Info. Link]
A visible light induced palladium-catalyzed fluoroalkylation method was developed. The Heck-type alkyl coupling reaction enables the introduction of trifluoroethyl, difluoroethyl and other fluoroalkyl fragment into styrenes under mild reaction conditions without the use of additional photosensitizers and ensures access to fluoroalkylated olefins on a broad scale.
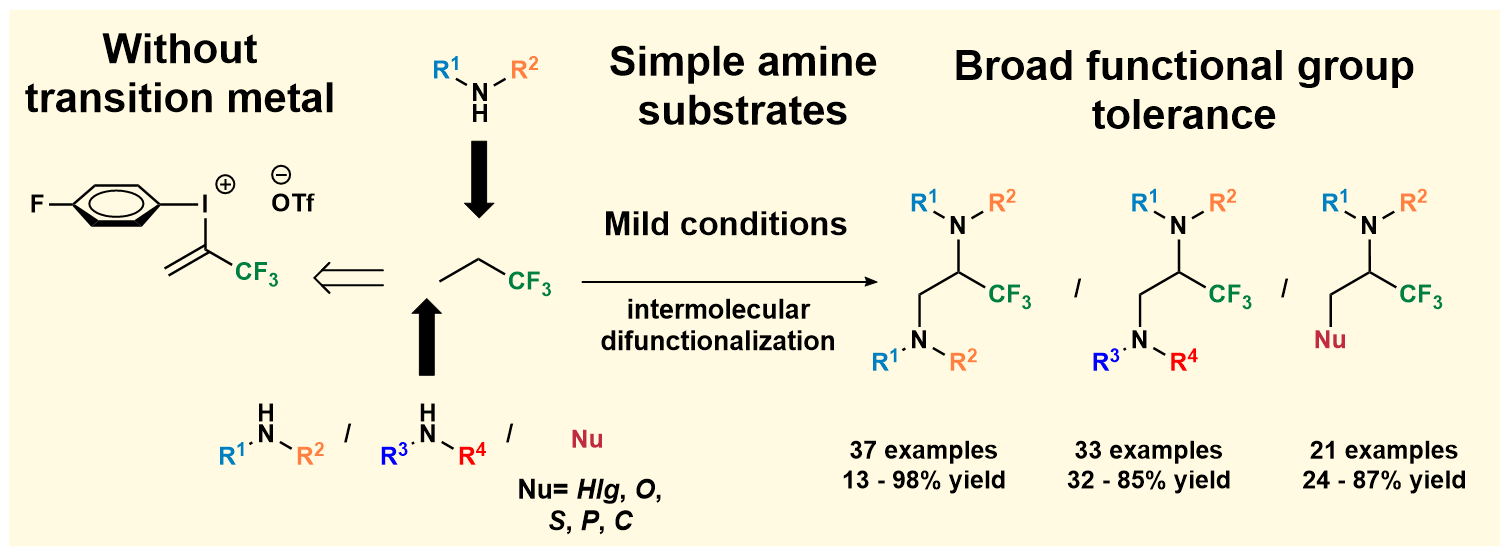
78. Vicinal difunctionalization of carbon-carbon double bond for the platform synthesis of trifluoroalkyl amines
Vicinal difunctionalization of carbon–carbon double bond for the platform synthesis of trifluoroalkyl amines, Ferenc Béke, Ádám Mészáros, Ágnes Tóth, Bence Béla Botlik, Zoltán Novák, Nat. Commun. 2020, 5924. DOI: 10.1038/s41467-020-19748-z | [Full Text Link] [Supp. Info. Link]
Regioselective vicinal diamination of carbon–carbon double bonds with two different amines is a synthetic challenge under transition metal-free conditions, especially for the synthesis of trifluoromethylated amines. However, the synthesis of ethylene diamines and fluorinated amine compounds is demanded, especially in the pharmaceutical sector. Herein, we demonstrate that the controllable double nucleophilic functionalization of an activated alkene synthon originated from a trifluoropropenyliodonium salt with two distinct nucleophiles enables the selective synthesis of trifluoromethylated ethylene amines and diamines on broad scale with high efficiency under mild reaction conditions. Considering the chemical nature of the reactants, our synthetic concept methodology brings forth an efficient methodology and provides versatile access to highly fluorinated amines.
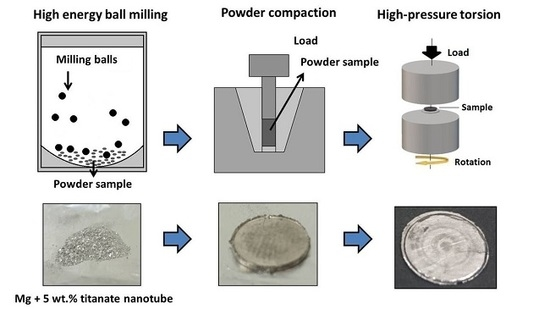
75. Microstructural Investigation of Nanocrystalline Hydrogen-Storing Mg-Titanate Nanotube Composites Processed by High-Pressure Torsion
Microstructural Investigation of Nanocrystalline Hydrogen-Storing Mg-Titanate Nanotube Composites Processed by High-Pressure Torsion, Marcell Gajdics, Tony Spassov, Viktória Kovács Kis, Ferenc Béke, Zoltán Novák, Erhard Schafler, Ádám Révész, Energies 2020, 13, 563. DOI: 10.3390/en13030563 | [Full Text Link]
A high-energy ball milling and subsequent high-pressure torsion method was applied to synthesize nanocrystalline magnesium samples catalyzed by TiO2 or titanate nanotubes. The microstructure of the as-milled powders and the torqued bulk disks was characterized by X-ray diffraction. The recorded diffractograms have been evaluated by the convolutional multiple whole profile fitting algorithm, which provided microstructural parameters (average crystal size, crystallite size distribution, average dislocation density). The morphology of the nanotube-containing disks has been examined by high-resolution transmission electron microscopy. The effect of the different additives and preparation conditions on the hydrogen absorption behavior was investigated in a Sieverts’-type apparatus. It was found that the ball-milling route has a prominent effect on the dispersion and morphology of the titanate nanotubes, and the absorption capability of the Mg-based composite is highly dependent on these features.
Keywords: ball milling; high-pressure torsion; magnesium; titanate nanotubes; hydrogen storage
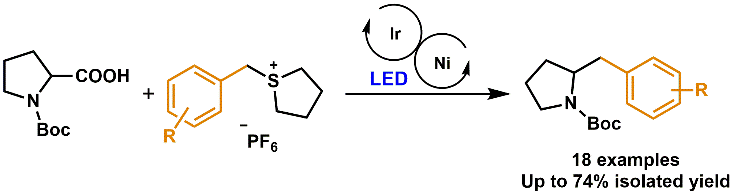
74. Development of Ni‐Ir dual photocatalytic Liebeskind coupling of sulfonium salts for the synthesis of 2‐benzylpyrrolidines
Development of Ni‐Ir dual photocatalytic Liebeskind coupling of sulfonium salts for the synthesis of 2‐benzylpyrrolidines, Bálint Varga, Zsombor Gonda, Balázs L. Tóth, András Kotschy, Zoltán Novák, Eur. J. Org. Chem. 2020. 10, 1466-1471. DOI: 10.1002/ejoc.201900957 | [Full Text Link] [Supp. Info. Link]
A new method has been developed for the synthesis of 2‐benzylpyrrolidines utilizing cross‐coupling and photoredox catalysis. Using a well‐established dual Ni‐Ir system, we were able to successfully couple benzylsulfonium salts with a proline utilizing radical forming through CO2 extrusion. This enabled the simple one step synthesis of 2‐benzylpyrrolidines from stable inexpensive starting materials.